Why Is The Phosphorus Cycle Important To Life
Phosphorus is a crucial element to life, whether an organism is a member of the plant or animal kingdom. It forms a fundamental component of genes – the DNA and RNA structure that determines what we are – and it also plays a major role in the ATP (adenosine triphosphate) energy cycle, without which we wouldn’t be able to contract our muscles. In vertebrates like us mammals, around 85 per cent of the phosphorus in our bodies can be found in our teeth and bones.
Phosphorus goes through a cycle similar to that of carbon, nitrogen and sulphur.
However, unlike these important systems, because of the Earth’s normal range of temperatures and pressures, hardly any of the phosphorus on our planet exists as a gas.
Instead, most of it is bound up in sedimentary rock and a small proportion in water, although phosphorus isn’t very soluble in H2O and tends to bond more readily to molecules in the soil, entering watery ecosystems as part of runoff particles.
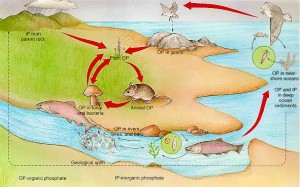 Phosphorous minerals, called phosphates, enter the food chain from rocks via weathering. Plants absorb the phosphorus ions in the soil, herbivores ingest phosphorus by eating the plants and, in turn, carnivores absorb it from herbivores. It’s then returned to the cycle via excretion and decomposition. Fertilizers, sewage and, formerly, detergents can all create an excess of phosphorus in the cycle, which can cause ‘blooms’ of suffocating algae and choking weeds in the sea and other bodies of water.
Phosphorous minerals, called phosphates, enter the food chain from rocks via weathering. Plants absorb the phosphorus ions in the soil, herbivores ingest phosphorus by eating the plants and, in turn, carnivores absorb it from herbivores. It’s then returned to the cycle via excretion and decomposition. Fertilizers, sewage and, formerly, detergents can all create an excess of phosphorus in the cycle, which can cause ‘blooms’ of suffocating algae and choking weeds in the sea and other bodies of water.
A dangerous gas
The gaseous form of phosphorus – phosphine – is usually only found under lab conditions as hydrogen phosphide (PH3) and is completely odourless in its pure form, although it has a strong rancid fish or garlic smell in its impure diphosphane form. Phosphine is also extremely flammable and toxic; concentrations as low as one part per million can quickly cause a number of short-term symptoms, including vomiting and breathing difficulties. Higher concentrations can cause permanent damage and even death. It does have a use in industry, however, playing a role in the manufacture of semiconductors (components vital to the electronics field) and also in pest control. In the latter it’s found as a gaseous fumigant or as phosphide pellets, treated to prevent the gas from exploding, which kill pests like rodents when inhaled/consumed.
Discovery of phosphorus
 Phosphorus was first discovered as an element in the 17th century. It was the 13th element to be found but the first since bismuth was discovered in ancient times. In 1669, German alchemist Hennig Brand came upon phosphorus when experimenting with urine in pursuit of creating the fabled philosopher’s stone – the substance that was widely thought to facilitate the transmutation of metals like lead into gold or silver.
Phosphorus was first discovered as an element in the 17th century. It was the 13th element to be found but the first since bismuth was discovered in ancient times. In 1669, German alchemist Hennig Brand came upon phosphorus when experimenting with urine in pursuit of creating the fabled philosopher’s stone – the substance that was widely thought to facilitate the transmutation of metals like lead into gold or silver.
Brand boiled litres of urine down into a paste that he heated through water, allowing its vapours to condense. Instead of gold, he got a waxy white substance that glowed in the dark: a phosphorous compound called ammonium sodium hydrogen phosphate. The glow that comes from white phosphorus is a slow reaction with oxygen that takes place at the surface of the element, which creates molecules that emit a visible wavelength of light. White phosphorus was used in matches for a time before it was removed because of its toxicity.

