Does Malaria Ever Go Away
Without drugs your chances of surviving severe malaria are nil. With drugs you should make it but, worryingly, even many medicines are losing their effectiveness.
“Drug resistance is a major threat. We know from history that every time we think we have outsmarted malaria, we learn the humbling lesson that we are dealing with a very wily parasite,” says Dr Robert Newman, director of the World Health Organization’s (WHO) global malaria programme.
However, on 25 April 2012, World Malaria Day, there were many things to celebrate. Deaths from malaria had fallen more than 25 per cent over the last decade. For the first time ever, a malaria vaccine had reached the final stage of clinical trials. There were also a number of promising new weapons being developed to fight mosquitoes, ranging from lasers that zap the bugs mid-flight, to fungi that disable the malaria parasite.
“We have an incredible opportunity ahead of us,” says Robert Newman. “Through universal coverage with current methods, and with the new tools in development, we can save millions of lives in the near term, and may be able to eradicate malaria in the next 40 years.”
A Global problem
Today, around half of the world’s population are at risk of malaria, with sub-Saharan Africa being one of the worst affected regions. Over half a million people die from malaria each year, and more than two million become ill. ‘Airport malaria’ – malaria transmitted by stowaway mosquitoes — is expected to become more of a problem as global temperatures rise.
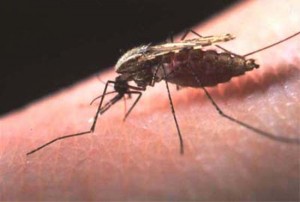
 Malaria is caused by a parasite called Plasmodium. These ingenious free-loaders live in the salivary glands of the Anopheles mosquito. As the mosquito bites, it spits out saliva, transferring hundreds of parasites into the human victim’s blood. From there, the parasites hitch a ride down to the liver, where they set up home inside a liver cell, growing and multiplying over the following week. Once matured, as many as one million Plasmodium may burst forth, invading the body’s red blood cells and clogging up the flow of oxygen to vital organs.
Malaria is caused by a parasite called Plasmodium. These ingenious free-loaders live in the salivary glands of the Anopheles mosquito. As the mosquito bites, it spits out saliva, transferring hundreds of parasites into the human victim’s blood. From there, the parasites hitch a ride down to the liver, where they set up home inside a liver cell, growing and multiplying over the following week. Once matured, as many as one million Plasmodium may burst forth, invading the body’s red blood cells and clogging up the flow of oxygen to vital organs.
The tiny, single-celled parasite is a master of disguise. By altering the proteins on its surface, Plasmodium is able to change its appearance regularly to avoid being recognized by our immune system. And its 5,000 genes are constantly mutating, developing resistance to all the drugs we throw at it. As a result, malaria was once considered too hard to beat.
But now two groups of researchers are getting tantalizingly close to producing a vaccine. The first of these vaccines is based on a strange phenomenon that was discovered in the 1970s. Researchers noticed that if mosquitoes were exposed to a blast of radiation, it damaged the DNA of Plasmodium in the mosquitoes’ salivary glands. “People who were bitten by these irradiated mosquitoes developed immunity to malaria,” explains Dr Stephen Hoffman, chief executive of Sanaria, a biotech company dedicated to creating a malaria vaccine.
 Radiation stops the parasites from maturing normally. This prevents them from using the shape-shifting ability that enables them to evade their host’s immune system. And because they stay the same, our bodies are able to recognize the parasites as a foreign invader and develop an immune response.
Radiation stops the parasites from maturing normally. This prevents them from using the shape-shifting ability that enables them to evade their host’s immune system. And because they stay the same, our bodies are able to recognize the parasites as a foreign invader and develop an immune response.
For the last decade, Stephen Hoffman has been trying to mimic this effect and create a vaccine, using Plasmodium extracted from the salivary juice of irradiated mosquitoes. The company started in Hoffman’s kitchen, but these days it has labs and US headquarters in Rockville, Maryland, and a team of highly-skilled staff — all proficient at dissecting mosquito saliva glands. “One person can dissect around 100 in one hour,” says Hoffman.
Results from its first clinical trial last year, in which 80 human volunteers were injected with the irradiated Plasmodium, were disappointing: just over five per cent of people achieved immunity. But a follow-up study in mice and monkeys resulted in over 70 per cent immunity, when it was injected directly into a vein. So Hoffman’s team is now conducting another clinical trial in humans, administering the vaccine intravenously.
Meanwhile, over at the GlaxoSmithKline labs in Belgium, Joe Cohen and his team are very close to producing a malaria vaccine too. Their potion, called Mosquirix, contains a fragment of Plasmodium from its blood-surfing stage inside the human body. When given the vaccine, our bodies learn to recognise the disguise worn by the parasite when it first enters, allowing us to arm our immune system ready to attack when the real thing arrives. “It only takes 30 minutes for Plasmodium to travel to the liver, so the body’s defences have to act very quickly,” says Dr Allan Pamba, a member of Cohen’s team.
 Preliminary results from phase III clinical trials (the final test) in over 15,000 young children look promising so far, cutting malaria rates by over 50 per cent. The trials are ongoing but, assuming no complications, WHO has said that Mosquirix could be licensed as soon as 2015.
Preliminary results from phase III clinical trials (the final test) in over 15,000 young children look promising so far, cutting malaria rates by over 50 per cent. The trials are ongoing but, assuming no complications, WHO has said that Mosquirix could be licensed as soon as 2015.
“We are thrilled by these results – this vaccine could save hundreds of thousands of lives every year,” says Pamba.
A vaccine that confers 50 per cent immunity still leaves a lot of people vulnerable, however, and effective treatment would require more than just drugs. Currently, for example, malaria is often misdiagnosed because the initial symptoms are similar to a really bad case of the flu: fever, chills, headache, sickness and muscle pain.
Mobilising treatment
But if you’re infected by the most deadly species of the parasite -Plasmodium falciparum – the disease can progress exceedingly rapidly, causing death within days. Diagnosing and treating malaria swiftly is clearly key to saving lives -and one tool that could help tackle the disease is the humble mobile phone.
Until now, many countries have used a paper-based system to track their medical supplies. In countries such as Tanzania, in East Africa, this has meant that around 40 per cent of clinics were out of malaria diagnosis kits and medicine at any one time, with new supplies sometimes taking months to arrive.
Modern gadgets could overcome this problem. A pilot programme in Tanzania (backed by IBM, Novartis and Vodafone) is thought to have saved hundreds of lives in just a few months last year, thanks to text messages, Google Maps and cloud computing software to track inventories of life-saving medications at remote clinics. It ensures that supplies are restocked before they run low. Such technological innovations, along with novel ways to tackle mosquitoes and new vaccines, will improve the lives of millions.
 Ending malaria will come at considerable financial cost — GlaxoSmithKline has already spent USD 300 million developing its vaccine over 25 years – but fortunately wealthy benefactors are committed to defeating the disease. Much of the progress on Mosquirix wouldn’t have been possible without the philanthropy of the Bill & Melinda Gates Foundation, which donated USD 200m to funding research. And with another USD 50-100m from GlaxoSmithKline, the project will be complete.
Ending malaria will come at considerable financial cost — GlaxoSmithKline has already spent USD 300 million developing its vaccine over 25 years – but fortunately wealthy benefactors are committed to defeating the disease. Much of the progress on Mosquirix wouldn’t have been possible without the philanthropy of the Bill & Melinda Gates Foundation, which donated USD 200m to funding research. And with another USD 50-100m from GlaxoSmithKline, the project will be complete.
Speaking at the Malaria Forum in October 2011, Bill Gates said, “Eventually, with relentless focus, we can eradicate malaria. We’ve already shrunk the malaria map considerably. We can make it disappear.” Excitingly, it looks like he could be right.
How to stop transmission of the malaria parasite
GENETIC MODIFICATION
One way of stopping malaria is to help mosquitoes fight off the Plasmodium parasite. This is exactly what George Dimopoulos, from John Hopkins University in Maryland, is trying to do.
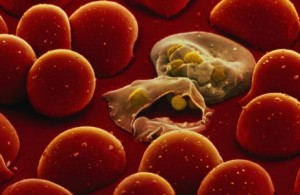 Last year Dimopoulos’s team managed to genetically modify the immune system of an Anopheles mosquito (pictured), making it highly resilient to the Plasmodium falciparum parasite. They took one of the insect’s own genes, and altered it so that the mosquito produced a faster and stronger immune attack when it fed on human blood. Studies in the lab showed that the GM mosquitoes became highly effective killing machines, preventing almost all Plasmodium parasites from taking up residence in their gut. Eventually, Dimopoulos envisages releasing a handful of the GM mosquitoes that would breed with their wild cousins – passing on their superior immune system with its malaria-resistant traits – in just a few generations.
Last year Dimopoulos’s team managed to genetically modify the immune system of an Anopheles mosquito (pictured), making it highly resilient to the Plasmodium falciparum parasite. They took one of the insect’s own genes, and altered it so that the mosquito produced a faster and stronger immune attack when it fed on human blood. Studies in the lab showed that the GM mosquitoes became highly effective killing machines, preventing almost all Plasmodium parasites from taking up residence in their gut. Eventually, Dimopoulos envisages releasing a handful of the GM mosquitoes that would breed with their wild cousins – passing on their superior immune system with its malaria-resistant traits – in just a few generations.
Malaria is carried by over 40 different species of mosquito, however, and genetically engineering every species isn’t feasible. Instead, the researchers aim to use the GM insect in regions where there’s only one predominant species of malaria-carrying mosquito.
NOSE BLOCKING
 Mosquitoes track down their victims by smell – homing in on odours of carbon dioxide and sweat. Now scientists are exploiting this fact, developing a chemical to block mosquitoes’ sense of smell. Without their powerful ‘nose’, mosquitoes will be left milling around aimlessly, unable to find their next meal. The first stage has been to identify the odour receptors that the mosquito uses to sniff out its human prey.
Mosquitoes track down their victims by smell – homing in on odours of carbon dioxide and sweat. Now scientists are exploiting this fact, developing a chemical to block mosquitoes’ sense of smell. Without their powerful ‘nose’, mosquitoes will be left milling around aimlessly, unable to find their next meal. The first stage has been to identify the odour receptors that the mosquito uses to sniff out its human prey.
Filippo Mancia from Columbia University and Vanessa Ruta from the Rockefeller University, both in New York, are using X-ray crystallography to reveal the atomic structure of the odour receptors on a mosquito’s antenna. After mapping out the exact shape and structure of the odour receptor, the scientists will then use Bayer CropScience’s database to search for chemicals with a mirror-image shape. These molecular mimics can latch onto the odour receptors, blocking the mosquito’s ‘nose’.
The end result will be the world’s most effective insect repellent. “Ultimately we should be able to develop something that will prevent any insect bite, which could be used to tackle not just malaria, but all insect-borne diseases,” says Dr Mancia.
KILLER FUNGUS
Mosquitoes are vulnerable to some types of fungus. Now researchers are taking advantage of this to genetically engineer a special fungus, designed to grab onto mosquitoes and block the exit route for Plasmodium falciparum – the most deadly form of the malaria parasite.
 The specially souped-up fungus contains a protein that prevents the parasite from entering the mosquitoes’ saliva glands, and an antibody to knock out the parasite. “Crucially, our fungus doesn’t stop the insects from being able to breed, which should prevent their evolving a resistance to it,” says Prof Raymond St Leger, from the University of Maryland. If the fungus did stop mosquitoes from breeding, it would trigger the insect’s immune system to develop a resistance to the fungus – preventing the fungal proteins from attacking the malaria parasite.
The specially souped-up fungus contains a protein that prevents the parasite from entering the mosquitoes’ saliva glands, and an antibody to knock out the parasite. “Crucially, our fungus doesn’t stop the insects from being able to breed, which should prevent their evolving a resistance to it,” says Prof Raymond St Leger, from the University of Maryland. If the fungus did stop mosquitoes from breeding, it would trigger the insect’s immune system to develop a resistance to the fungus – preventing the fungal proteins from attacking the malaria parasite.
In lab-based tests, Plasmodium was nearly completely eradicated from the saliva glands of mosquitoes dosed with the fungus. The researchers are now arranging permits to set up field trials in Burkina Faso, West Africa. They plan to construct giant domes covered in fungus-impregnated net, with the aim of creating a malaria-free zone inside the dome. As well as protecting humans, it could also protect livestock.
ZAP ‘EM WITH LASERS
If all else fails, we could resort to frying mosquitoes using laser beams. Intellectual Ventures, the company belonging to Nathan Myhrvold, Microsoft’s former chief technology officer, has already made a prototype – cobbled together from parts found in printers, digital cameras and projectors.
 In a public demonstration at the 2010 TED conference in Long Beach, California, Myhrvold used the machine to gun down hundreds of mosquitoes contained inside a glass tank. The laser tracked the movements of the mosquitoes and then shot them down, with each mosquito going up in a plume of smoke. The team say that their device can shoot down between 50 and 100 mosquitoes per second.
In a public demonstration at the 2010 TED conference in Long Beach, California, Myhrvold used the machine to gun down hundreds of mosquitoes contained inside a glass tank. The laser tracked the movements of the mosquitoes and then shot them down, with each mosquito going up in a plume of smoke. The team say that their device can shoot down between 50 and 100 mosquitoes per second.
Luckily the laser software is sophisticated enough to recognise its prey before shooting, preventing it from cooking butterflies, for example. It can even differentiate between male and female mosquitoes, ensuring that only the malaria-carrying females are killed. Myhrvold envisages using the laser to create protective ‘fences’ around people’s houses, doctor’s clinics, or perhaps even agricultural fields. They estimate that such a device could cost as little as USD 50.

